Abstract
Introduction Tyrosine kinase inhibitors (TKIs) are commonly used as first-line therapy for the treatment of patients with chronic myeloid leukemia in the chronic phase (CML-CP). Molecular response is monitored regularly after initiating TKI therapy and less frequently after achieving BCR::ABL1 ≤ 0.1%. Although responses to TKIs have been extensively evaluated in clinical trials, utilization of molecular testing and clinical response associated with TKI therapy is not as well understood in real-world settings. SIMPLICITY (NCT01244750) is a prospective observational study assessing TKI use, molecular testing patterns, and clinical response in patients with CML-CP in routine clinical practice.
Methods The SIMPLICITY study followed patients with CML-CP from routine clinical practice who received first-line treatment with the first-generation TKI imatinib (IM) or second-generation TKIs dasatinib (DAS) or nilotinib (NIL) in the USA and Europe. As this was an observational study, molecular testing was performed at the discretion of the treating team without directives from the study protocol. The cumulative proportion of US patients who underwent ≥ 1 molecular test as well as the rates of major molecular response (MMR; BCR::ABL1 ≤ 0.1%) and deep (4.5-log) molecular response (MR4.5; BCR::ABL1 < 0.0032%) were evaluated through 3 and 6 months, and yearly through 5 years, after starting TKI treatment. Patients were stratified by index TKI (IM or DAS/NIL). Descriptive statistics are presented.
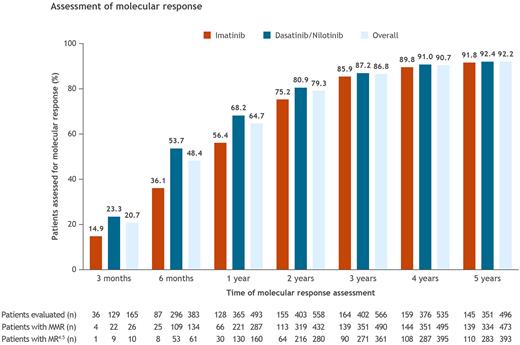
Results At data cutoff (January 28, 2020), a total of 810 prospective US patients were enrolled and included in this analysis, of whom 244 received IM and 566 received DAS/NIL. The median age (range) at diagnosis was 56 (18-91) years (IM, 58 [18-90] years; DAS/NIL, 55 [19-91] years). A total of 796 (98.3%) patients were followed through 3 months from the start of index TKI treatment, 792 (97.8%) through 6 months, 762 (94.1%) through 1 year, 704 (86.9%) through 2 years, 652 (80.5%) through 3 years, 590 (72.8%) through 4 years, and 538 (66.4%) through 5 years. Of those followed, the overall proportion of patients tested for molecular response ranged from 20.7% through 3 months to 92.2% through 5 years (Figure). The proportion of assessed patients who achieved MMR at least once increased over time; MMR was achieved in 15.8% of patients assessed through 3 months (IM, 11.1%; DAS/NIL, 17.1%), 35.0% through 6 months (IM, 28.7%; DAS/NIL, 36.8%), 58.2% through 1 year (IM, 51.6%; DAS/NIL, 60.5%), 77.4% through 2 years (IM, 72.9%; DAS/NIL, 79.2%), 86.6% through 3 years (IM, 84.8%; DAS/NIL, 87.3%), 92.5% through 4 years (IM, 90.6%; DAS/NIL, 93.4%), and 95.4% through 5 years (IM, 95.9%; DAS/NIL, 95.2%). The proportion of patients who achieved MR4.5 at least once also increased over time; MR4.5 was achieved in 6.1% of patients assessed through 3 months (IM, 2.8%; DAS/NIL, 7.0%), 15.9% through 6 months (IM, 9.2%; DAS/NIL, 17.9%), 32.5% through 1 year (IM, 23.4%; DAS/NIL, 35.6%), 50.2% through 2 years (IM, 41.3%; DAS/NIL, 53.6%), 63.8% through 3 years (IM, 54.9%; DAS/NIL, 67.4%), 73.8% through 4 years (IM, 67.9%; DAS/NIL, 76.3%), and 79.2% through 5 years (IM, 75.9%; DAS/NIL, 80.6%). Among all 810 enrolled patients, 473 (58.4%) and 393 (48.5%) achieved MMR and MR4.5, respectively, and remained in the study through 5 years. Mean (standard deviation) time from index TKI to best response in tested patients was 2.4 (0.5) months among patients assessed through 3 months and 24.4 (15.4) months among those assessed through 5 years.
Conclusions At 5-year follow-up in US patients from the SIMPLICITY study, there was a consistent increase in the proportion of assessed patients who achieved MMR and MR4.5 over time for all patients treated with TKIs, with the caveat that patients who remained on treatment at each subsequent time point were more likely to respond. In general, a consistently higher proportion of patients treated with DAS/NIL achieved MR4.5 across the time points assessed compared with IM in this non-controlled study. Further studies evaluating molecular response to IM, DAS, NIL, and other TKIs beyond 5 years can help delineate further differences in outcomes and determine whether molecular testing frequency correlates with outcomes.
Study support This study was funded by Bristol Myers Squibb.
Disclosures
Cortes:Forma Therapeutic: Consultancy; Gilead: Consultancy; Biopath Holdings Inc: Consultancy, Current equity holder in private company; Abbvie: Consultancy, Research Funding; Pfizer: Consultancy, Honoraria, Research Funding; Takeda: Consultancy, Honoraria; Sun Pharma: Consultancy, Research Funding; Novartis: Consultancy, Honoraria, Research Funding. Goldberg:COTA Healthcare: Current equity holder in private company, Current holder of stock options in a privately-held company; MDS Foundation: Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy. Paquette:Novartis: Consultancy. Tang:Bristol Myers Squibb: Current Employment, Current equity holder in publicly-traded company. DeGutis:Bristol Myers Squibb: Current Employment, Current equity holder in private company, Current holder of stock options in a privately-held company. McBride:Bristol Myers Squibb: Current Employment. Parsons:ICON Clinical Research: Current Employment. Montelongo:ICON plc: Current Employment. Mauro:Sun Pharma/SPARC: Research Funding; AbbVie, Bristol Myers Squibb, Novartis, Pfizer, Takeda: Consultancy, Honoraria, Other: Travel, accommodation, expenses , Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal